
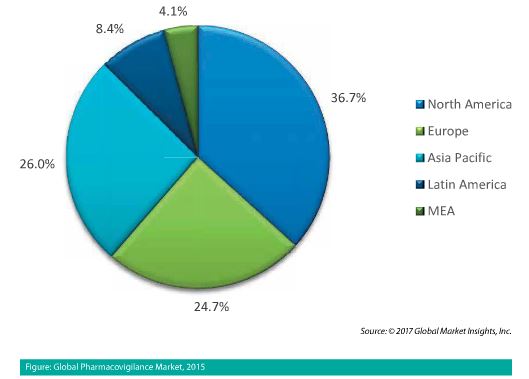
The pharmacovigilance market dynamic
pharmafile | August 25, 2017 | News story | Medical Communications | biotech, drugs, pharma, pharmaceutical, pharmacoviglance
Pharmacovigilance is a science of understanding drug safety through identification, assessment, and prevention of adverse drug reactions (ADRs). It is an ongoing approach encompassing preclinical, clinical and post-clinical drug testing. Pharmacovigilance services mainly include drug safety assessment and adverse drug monitoring services at different clinical trial stages including post marketing surveillance.
Clinical trials are conducted to gather safety and efficacy data for a new drug entity. Preclinical trials are early stage drug studies supporting the later studies on human trials. Phase 1 trials identifies appropriate therapeutic dose by assessing the safety, tolerability, pharmacokinetics, and pharmacodynamics of a drug on patients. Phase 2 trials assess the mechanisms and effectiveness of drug. Phase 3 studies helps determine drug effectiveness compared to the existing gold standard therapy. It is the most expensive, time consuming and difficult phase to plan and implement out of all other phases. Phase 4 is the post-commercialisation studies operational throughout the product lifecycle carried out to identify any long-term adverse effects over a larger patient pool. Adverse drug events detected during Phase 4 trials may potentially result into market withdrawal, or territorial marketing limitations.
Pharmacovigilance is proving a cost-effective solution to patients as well as healthcare institutions by preserving the safety and quality of human lives. Increasing number of drug approvals and growing polypharmacy trends should contribute to escalated instances of ADRs among patients. High-risk patient pool requires a great degree of attention for detection and monitoring of adverse drug events.
Reporting of ADRs leads to increased drug awareness and can help prevent its occurrence in outpatient as well as inpatient settings, eventually reducing hospital admissions and readmissions. Pharmacovigilance can also allow medical professionals to use their discretion while choosing a drug and its dosage.
Stringent drug safety regulations have made drug manufacturers to comply with numerous pharmacovigilance protocols and maintain their market authorisation to commercialise their products. The US FDA primarily focuses on drug safety profile during clinical development stages and requires the submission of reports for serious adverse effects. In the EU, Good Pharmacovigilance Practices (GVP) apply to all authorised medical products. The European Medicines Agency (EMA) requires all serious adverse drug events to be submitted within 15 calendar days, and all non-serious drug events within 90 calendar days by the applicant.
The global pharmacovigilance market will be driven by rising disease burden with sedentary lifestyles, constant developments and sophistication in healthcare services, innovative therapeutics, and development better and safer medications. High R&D expenditure, increasing number of ADRs due to improved awareness levels and rise in drug consumption should spur pharmacovigilance market growth.
Developing economies are growing at much faster rate and provides excellent platforms for future market growth due to increasing population, rising healthcare expenditure, improved healthcare infrastructure and presence of untapped opportunities.
Pharmacovigilance outsourcing has grown massively over the past decade. The outsourcing of pharmacovigilance tasks provides a flexible solution to the increasing demands. Moreover, the companies are looking for options to reduce costs for maximising their R&D productivity. With the growing drug consumption, the need for regular monitoring of drugs has also augmented, eventually boosting the pharmacovigilance market.
Major factors impeding pharmacovigilance market growth are online sales of medicines, increasing self-medication, easy access to drug information and polypharmacy. Direct marketing to the consumer of prescription drugs has become ordinary in many nations. Communication between national pharmacovigilance centres and national drug regulatory institutions should be improved for rapid and effective regulatory decisions.
The pharmacovigilance market is highly competitive with many small, large, and global players operating in the industry space. Some of the key industry participants include Accenture, Boehringer Ingelheim, Bristol-Myers Squibb, Covance, Icon, Parexel, Quintiles, Cognizant Technology Solutions Corporation, and United BioSource. The major tactics adopted by industry players include strategic collaborations with contract organisations, mergers and acquisitions and geographical expansion through partnerships and integrations.
Related Content

Arkin Capital closes $100m fund for pre-clinical and early clinical-stage biotech
Arkin Capital has announced the closing of Arkin Bio Ventures III, a $100m fund designed …

Cellbyte raises $2.75m to fund pharma drug launch platform
Cellbyte has announced that it has raised $2.75m in seed funding for the streamlining of …

Lilly opens fourth US Gateway Labs site
Eli Lilly has opened its newest Lilly Gateway Labs (LGL) site in San Diego, California, …







