Kite Pharma says trial death was unrelated to KTE-C19
pharmafile | August 18, 2015 | News story | Medical Communications, Research and Development | CAR-T, cinical trials, kite pharma, non-Hodgkin's lymphoma, non-Hodgkin’s lymphoma
Kite has confirmed last week’s rumours that a patient had died during its Phase I/II trial for the KTE-C19 non-Hodgkin’s lymphoma treatment, while reassuring investors that the death was unrelated to the treatment.
Kite shares fell by more than 16% on Friday when the rumours circulated and a delay in the trial was considered a possibility, but have recovered since Kite CEO Arie Belldegrum reported the update to analysts.
“It is not now, nor has it ever been, on any type of clinical hold by the FDA or any other regulatory body,” Belldegrun said this week.
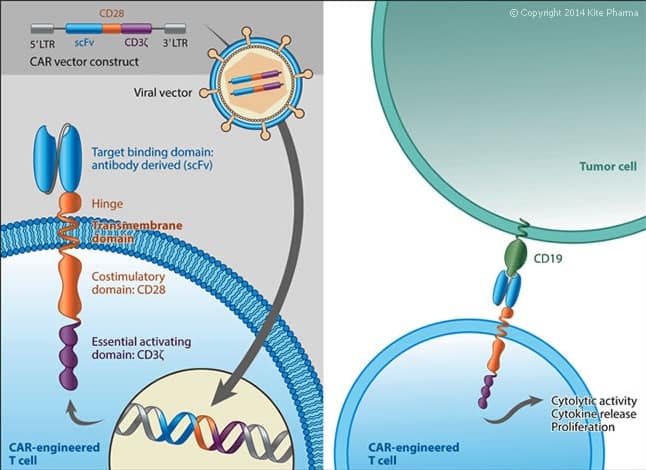
KTE-C19 is an investigational therapy in which a patient’s T cells are genetically modified to express a Chimeric Antigen Receptor (CAR) designed to target the antigen CD19, a protein expressed on the cell surface of B-cell lymphomas and leukemias.
The trial involves patients with refractory aggressive non-Hodgkin’s lymphoma (NHL) who have failed prior chemotherapy treatments and have a poor prognosis, meaning a death is not wholly unexpected.
Yet investors are nervous regarding CAR-T therapies, which are associated with cytokine release syndrome, an immune response that can be fatal to a patient.
Kite said multiple patients have been treated with KTE-C19 since the Phase 1 portion of the trial began in May, and complete responses have been observed by investigators, which are now being monitored to determine the treatment’s longevity. Belldegrun spoke of how the therapy had “melted away tumours,” during the trial.
The California-based company plans to present top-line data from the Phase 1 portion of the trial at the upcoming 2015 American Society of Hematology (ASH) Annual Meeting in December.
“We are encouraged by the progress of the KTE-C19 clinical trial and excited by the responses we have seen so far. We believe the KTE-C19 clinical findings are in line with previous results demonstrating the potential of this promising therapeutic approach,” says Belldegrun.
“In agreement with ASH, we have taken this exceptional step of providing an update on the trial in order to address recent misinformation in the market related to our clinical program. We are on track to transition to the Phase 2 portion of the trial and plan to present Phase 1 data at ASH later this year.”
Joel Levy
Related Content

European approval for Kite’s CAR-T therapy Tecartus in elapsed or refractory mantle cell lymphoma
Gilead-owned Kite Therapeutics has secured marketing authorisation from the European Commission for its chimeric antigen …

Novartis unveils new multi-trial data for CAR-T therapy Kymriah in advanced lymphoma
Novartis has taken the opportunity at the 2020 American Society of Hematology Annual Meeting & …

EMA’s CHMP recommends conditional approval for Kite Pharma’s CAR T therapy for mantle cell lymphoma
Kite Pharma’s anti-CD19 chimeric antigen receptor (CAR) T cell therapy known as KTE-X19 has received …






