French regulator probes Teva plant after patient death
pharmafile | June 12, 2013 | News story | Manufacturing and Production | Teva, furosemide, zopiclone
France’s medicines regulator has started an investigation into a manufacturing facility operated by generic firm Teva in the wake of a death that may have been caused by a packaging error.
The ANSM (Agence Nationale de Sécurité de Médicament et des Produits de Santé) launched the probe after the death of a 91-year-old man in Marseille, who is thought to have been taking a generic furosemide diuretic product that was subject to a Teva recall earlier this week.
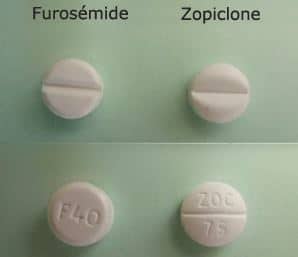
The recall of the Teva product was undertaken after it emerged that a production error could have resulted in substitution of furosemide tablets with zopiclone (see main image), a treatment for insomnia, in some blister packaging.
In a statement issued yesterday, the ANSM said it was “not possible to establish a causal link between the death of the patient and the packaging problem” on the basis of information currently available.
The agency notes however that substitution of the two medicines could have caused health risks to the patient, for example by reducing the protective diuretic effect of furosemide which is prescribed for high blood pressure and congestive heart failure.
Second death under scrutiny?
At the time of writing there were reports that another patient death was also being investigated for a possible link to the packaging error, although no official comment on that was available. Meanwhile, the ANSM had been due to carry out a site inspection at the affected Teva facility in Sens, Burgundy, yesterday.
The recall affected two lots of the furosemide product (Y175 and Y176, both expiring in 08/2015), which collectively equate to around 190,000 boxes of the medication.
The Conseil National de l’Ordre des Pharmaciens (CNOP) has ordered a ban on sale of the lots and advises patients which have the affected packs in their possession to return them to their pharmacist for replacement.
Teva is having a run of bad luck in its manufacturing network at the moment, having suffered an explosion at an active pharmaceutical ingredient (API) facility in Israel last week that resulted in one fatality and injured 30 workers.
Phil Taylor
Related Content

Teva’s Ajovy significantly reduces migraine in children and adolescents
Teva’s Ajovy (fremanezumab) has demonstrated significant reduction in monthly migraine and headache days in children …

Teva announces positive results from trial of AJOVY for migraine
Teva has announced positive results from the phase 4 PEARL study of AJOVY (fremanezumab), its …

Sanofi and Teva partner for development of inflammatory bowel disease treatment
Sanofi and Teva Pharmaceuticals have announced that they will collaborate for the co-development and co-commercialisation …






