$1.1 billion swoop for promising Parkinson’s drug developer
pharmafile | July 24, 2017 | News story | Sales and Marketing | Mitsubishi Tanabe Pharma Corporation, Neuroderm, biotech, drugs, pharma, pharmaceutical
Mitsubishi Tanabe Pharma Corporation (MTPC) has announced that it has moved to acquire Israeli pharma company, NeuroDerm, in a deal worth $1.1 billion. The deal sees MTPC gain access to NeuroDerm’s promising Parkinson’s disease treatment, ND0612, with the hope that approval could see it strengthen its stake in the US market.
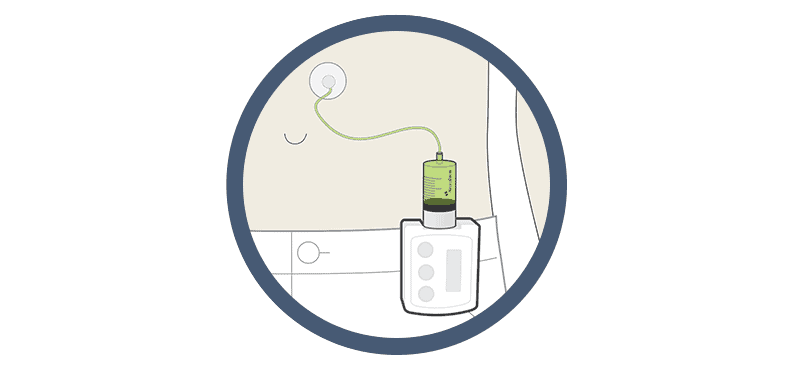
ND0612 has already received promising Phase 2 results where it performed strongly, and led to favourable comparisons beyond AbbVie’s Duodopa for the management of Parkinson’s. The product will be used to treat those who cannot control their motor problems with oral levodopa. ND0612, instead, will be delivered as a continuous dose over a 24-hour period by a belt pump (image above) or patch pump.
In Phase 2 trials, it was shown to have a strong safety profile and displayed improved usage. As the administration process is continuous, it also resulted in the reduction of fluctuation in the level of plasma levodopa concentrations in patients compared with placebo treatment.
The drug is now undergoing Phase 3 trials and MTPC mentioned that it believes the product will launch in 2019. In its statement explaining the decision to acquire NeuroDerm, MTPC stated: “the acquisition of ND0612 through this transaction is intended to enable MTPC to achieve its US sales of 80 billion yen by FY2020.”
For NeuroDerm, it represents one of the largest acquisitions of an Israeli firm and the $1.1 billion deal in cash represents a premium of 17% on stock prices on Friday.
“We believe that this transaction will yield important benefits for NeuroDerm’s shareholders and the Parkinson’s disease patients that urgently need new therapies,” said Oded S. Lieberman, CEO of NeuroDerm. “MTPC has demonstrated development and commercialization expertise in the field of neurology and we are confident that the combination of their resources and the robust data supporting ND0612, our Phase III Parkinson’s disease product candidate, will help make this important new therapy available as broadly and rapidly as possible.”
The deal is expected to close by the end of 2017.
Ben Hargreaves
Related Content

Arkin Capital closes $100m fund for pre-clinical and early clinical-stage biotech
Arkin Capital has announced the closing of Arkin Bio Ventures III, a $100m fund designed …

Cellbyte raises $2.75m to fund pharma drug launch platform
Cellbyte has announced that it has raised $2.75m in seed funding for the streamlining of …

Lilly opens fourth US Gateway Labs site
Eli Lilly has opened its newest Lilly Gateway Labs (LGL) site in San Diego, California, …






