
Navigating the challenges of health technology assessment
pharmafile | May 31, 2017 | Feature | Business Services, Manufacturing and Production, Medical Communications, Research and Development, Sales and Marketing | GPI, Global Pricing Innovations
In the face of restricted budgets, payers worldwide are increasingly using Health Technology Assessments (HTAs) as a way of managing healthcare budgets by providing access to therapies that demonstrate robust clinical and economic effectiveness.
HTAs are a systematic method of evaluating new technologies by considering the social, economic, organisational and ethical components of new health care interventions. At present, the European HTA system is incredibly fragmented with 77 HTA organisations spread across 29 European countries, each increasingly expanding the application of HTA evaluation. To add to the complexity of the situation, each HTA body has its own unique method of evaluation, resulting in varying submission requirements and access restrictions across countries.
HTA recommendations with negative outcomes have resulted in payers gaining the opportunity to negotiate and drive down treatment costs by demanding companies justify and support the value of the therapy. Companies, on the other hand, resort to bargaining for access through innovative price/access schemes, price reductions or resubmission with additional data. Submission rejections range from unacceptable incremental cost effectiveness ratios (ICERs) to insufficient evidence and incorrect comparator choice. The recommended comparator itself, varies drastically from country to country. In the case of Invokana, for example, the German HTA body IQWIG deemed the therapy to have “no added benefit” due to issues with comparator studies in the dossier. G-BA specified appropriate comparators for each of the 4 therapeutic indications which differed to the comparators submitted by the manufacturer. In contrast, the Canadian HTA body for Quebec, INESSS, found Invokana to have a similar efficacy to Sitagliptin (the comparator drug submitted in the dossier).
A key challenge for companies, therefore, is understanding the diverse and constantly changing evidence requirements and frameworks for each HTA agency across countries. The extent to which HTA outcomes vary across countries is illustrated by the case of Nexavar, indicated for renal cell carcinoma (Figure 1). Decisions differ dramatically across countries, from no recommended use in the UK, to positive recommendations in France and Sweden.
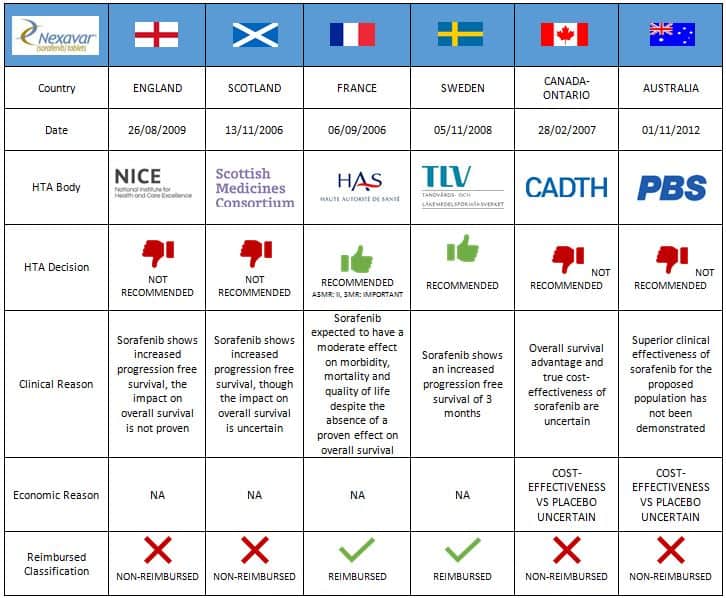
Source: GPI pulse™ – 1st March 2017
As HTA bodies become more powerful and assessments become a common way of restricting therapies at national and regional levels to maximize budgets, awareness of agency HTA requirements and payer perceptions is a crucial requirement for companies.
What can companies do? Optimise price and access strategies by leveraging data-driven insights
Companies can leverage historical outcomes of submissions to gain insights by learning from the past. Leveraging these insights can minimise the chance of unsuccessful outcomes such as rejected submissions, recommendations at a reduced-price point or restricted access. Considering the HTA information for renal cell carcinoma (RCC) in the table above, we can clearly see large differences between the evidence submitted by manufactures and those required by the agencies themselves leading to protracted and failed negotiations.
As HTAs gain more significance, a coherent strategy and awareness of payer requirements and perceptions can ensure that clinical trials are designed around the needs of payers, to maximise the likelihood of positive reimbursement/access decisions and reduce delays ensuring the best price and access outcome in a timely manner. Retrospective analysis of historical outcomes can help us understand payer thinking and by having access to real-time payer insights can help make better decisions by anticipating payer thinking. As HTA strategies continuously evolve, there is a need to address the current issues with HTA and what can be considered to overcome these issues and to minimise the time it takes for a new drug or therapy to come into the market.
GPI pulse™ is a new platform that provides timely intelligence helping companies optimise price and access strategies through real-time reimbursement, access and HTA insights. The innovative platform leverages the latest technology to combine multiple data-streams and analytics to provide necessary insights for successful market access decisions across countries. Contact info@globalpricing.com or visit www.globalpricing.com for further information.
By Pati Ladron, Global Pricing Innovations






